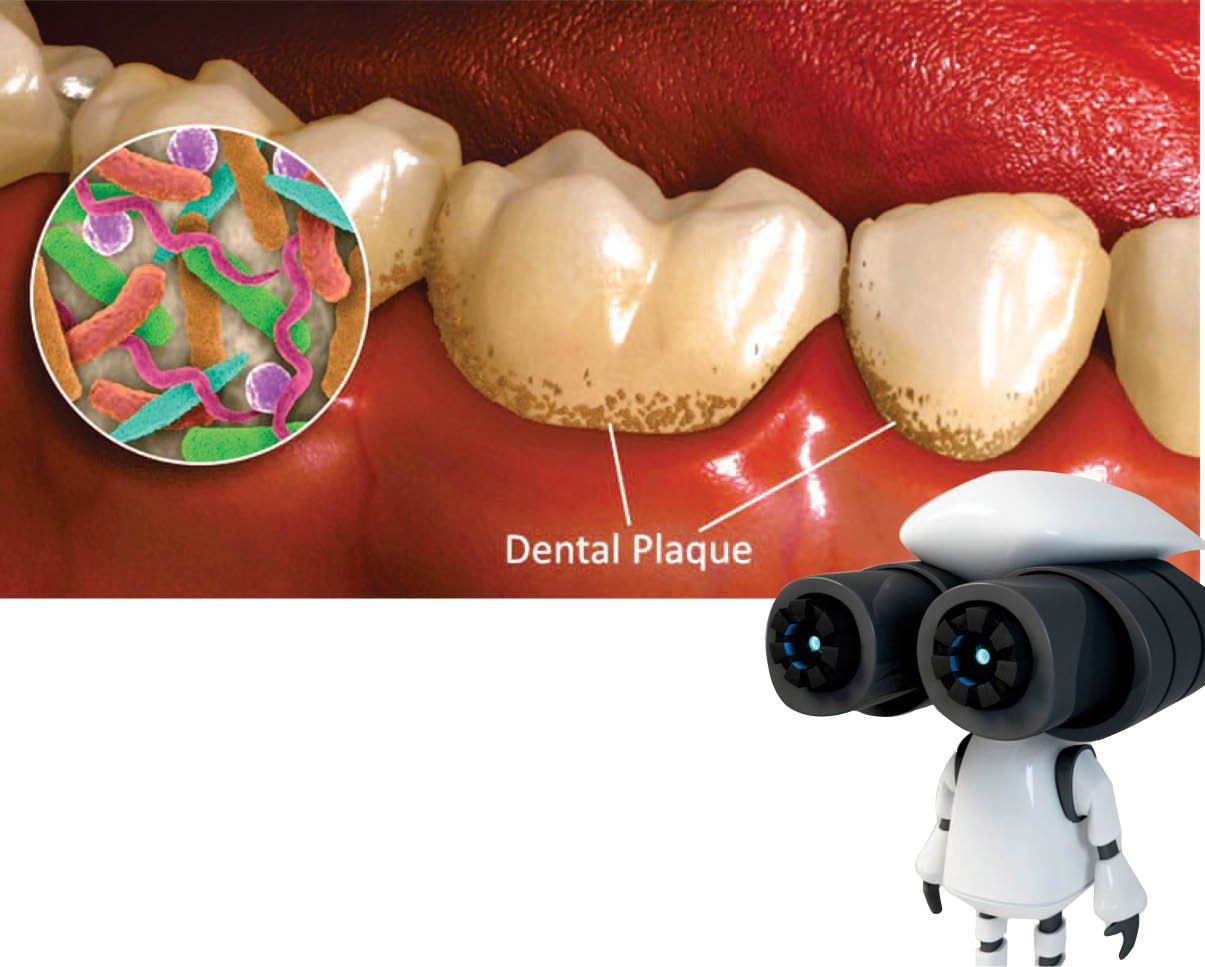
An adequate visit to the dentist typically consumes the time and some of the times it is such an unpleasant scraping with mechanical tools to remove plaque from teeth. What if, a dentist could station for a small army of tiny robots to precisely and non-invasively remove that buildup? In the University of Pennsylvania a team of engineers, dentists, and biologists developed a microscopic robotic cleaning crew. Whereas with two types of robotic systems — the one to operate inside confined spaces and the other one designed to work on surfaces the scientists showed that robots with catalytic activity could ably destroy biofilms, sticky amalgamations of bacteria enmeshed in a protective scaffolding. These robotic biofilm-removal systems are valuable in a wide range of capable applications, to reducing the risk of tooth decay, endodontic infections, and implant contamination from keeping water pipes and catheters clean.
Biofilms, they arise on biological surfaces, such as on an object or in a joint or on tooth, like implants, catheters, or water pipes. Wherever biofilms form, they are scandalously difficult to remove, as they are like a sticky matrix that holds the bacteria provides protection from antimicrobial agents. This work, is published in Science Robotics, was led by Hyun (Michel) Koo who is from the School of Dental Medicine and Edward Steager of the School of Engineering and Applied Science.
For the switch this innovation down to the clinical application, the researchers or the innovators are receiving support from the Penn Center of Health, Devices, and Technology, an initiative support by Penn’s Perelman School of Medicine, Penn Engineering, and the Office of the Vice Provost for Research. As its known, Penn health-tech awards select interdisciplinary groups with support to create new health technologies. By plowing away the degraded remains of the biofilm, the chance of it taking hold and re-growing decreases substantially. The robots are precisely directed by researchers to wherever they need to go to remove biofilms, be it the inside of a cathether or a water line or difficult-to-reach tooth surfaces.
In Koo’s previous work, Koo and colleagues have made headway at breaking down the biofilm matrix with a maximum variety of outside-the-box methods. One planning has to employ iron-oxide-containing nano particles that work catalytically, activated hydrogen peroxide to release free radicals that can kill bacteria and destroy biofilms in a targeted issue. The Penn Dental Medicine team searched that groups at Penn Engineering led by Steager, Vijay Kumar, and Kathleen Stebe were working with a robotic platform that used very similar iron-oxide nano particles as building blocks for microrobots. The engineers are able to control the movement of these microrobots using a magnetic field, allowing a tether-free way to steer them.
The cross-school team collaborates and they designed, optimized, and tested two types of robotic systems, these are the group that calls catalytic antimicrobial robots, or commonly CARs, they are capable of degrading and removing biofilms. This first involves switching iron-oxide nanoparticles in a solution, further that can be directed by magnets to remove biofilms on a surface in a plow-like manner. The second stage entails merge the nano particles into gel molds in three-dimensional shapes. These were used to aim the target and destroy biofilms clogging enclosed tubes. The CARs were functioned to degrade and remove bacterial biofilms not just from a tooth surface but from most difficult-to-access parts of a tooth, which are mostly the isthmus, which are a narrow corridor between root canals where biofilms commonly grow. The CARs effectively killed all the bacteria which broke down the matrix that surrounds them, and removed the debris with high quality. After testing the robots on bio-films growth on flat glass surface and enclosed glass tubes, the researchers tried out many more clinically relevant application: Removing biofilm from hard-to-reach parts of a human tooth.
Treating biofilms that occur on teeth requires a great deal of manual labor, both on the part of the consumer and the professional. The hope to improve treatment options as well as reduce the difficulty of care. The penn health-tech team has a great clinical background on the dental side and a great technical background on the engineering side. We’re here to round them out on the business side. They have really done a fantastic job on the project.
-Vaidehi Thakurw